Natron Energy, an American battery tech startup founded in 2012, develops high-performance rechargeable sodium-ion batteries thanks to their advancements in prussian blue electrodes and electrolytes. Their batteries store and transfer sodium-ions faster and with less internal resistance than any other commercially available batteries. With zero strain during charge/discharge, 10x faster cycling, and an over 50,000 cycle-life, Natron’s sodium-ion batteries represent the future of energy storage.
Challenges: lithium battery
Lithium-ion batteries have a high energy density, which means they can store more energy in a smaller and lighter package than other battery technologies. They have a low rate of self-discharge, which means they can keep their charge for longer.
The lithium-ion battery technology has reached maturity after many years of research and work to make it useful in industry. They have been used a lot in portable electronics like smartphones, laptops, and personal digital assistants. They have also been used in electric cars, electric forklifts, automated guided vehicles, boats, yachts, storing intermittent solar and wind energy, and giving backup power to critical equipment.
But making and throwing away lithium-ion batteries can be bad for the environment. For example, lithium and other metals have to be taken out of the ground, and a fire could release toxic chemicals. Lithium-ion batteries can be expensive, which keeps them from being used on a large scale to store energy. Also, several dozens of lithium-ion energy storage systems are said to have exploded or caught fire.
But making and throwing away lithium-ion batteries can have negative environmental impacts, such as the extraction of lithium and other metals, as well as the potential release of toxic chemicals in landfills. Lithium-ion batteries can be expensive, which potentially prevents them from widespread applications in grid-scale energy storage. Also, several dozens of lithium-ion energy storage systems are reported to have exploded or caught fire.
Therefore, low-cost, high-performance, and low fire risk battery technology is required for the future energy storage.
Natron Energy Technology
Natron Energy develops sodium-ion batteries utilizing inexpensive prussian blue materials and natural abundance of sodium. The open-framework nature of the cubic structure of prussian blue, which contains open channels and interstitial sites, enables rapid solid-state diffusion of sodium ions. Their sodium-ion batteries have a lower cost, a lower fire risk, rapid recharge capabilities, a wide operating temperature range, a longer cycling lifetime, and are more environmentally friendly than commercial lithium-ion batteries, making them a future energy storage alternative to lithium-ion batteries.
Natron sodium ion battery
The diagram below depicts the structure of the Natron sodium-ion battery. The battery consists of current collectors, prussian blue anode, prussian blue cathodes, and an electrolyte layer. Between the two electrodes, an insulating porous separator that facilitates ion transport of the electrolyte may also be present.
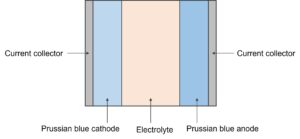
Symmetric battery configuration
Natron Energy’s batteries employ a symmetric configuration. Anode and cathode are composed of the same prussian blue material. Prussian blue has an open-framework nature of the cubic structure that enables rapid solid-state diffusion of sodium ions. Prussian blue has longer cycle life and can operate at higher charge/discharge rates than other asymmetric electrode systems, making the symmetric battery desirable. If one prussian blue electrode is paired with a different type of electrode, the battery may not last as long or provide the same high-rate capabilities as a symmetric cell containing a prussian blue anode and cathode.
Innovation of prussian blue materials
Natron Energy develops stable, high-performance prussian blue electrodes. The prussian blue material is a transition metal cyanide coordination compound. It can be represented generally as AₘPₙ[R(CN)6]ⱼ∙nH₂O, wherein A is an alkali metal cation such as Na⁺, P is at least one of manganese and iron, and R is at least one of manganese and iron.
The prussian blue of sodium manganese hexacyanomanganate Na₂Mn[Mn(CN)6]∙nH₂O has a capacity of over 200 mAh g⁻¹ that is surprisingly higher than other prussian blue materials, making it desirable for high-performance sodium-ion batteries. However, manganese hexacyanomanganate is air and moisture sensitive and thus its storage, handling, and processing require a controlled environment devoid of oxygen and moisture. Incorporating such a material into a battery product increases production cost and reduces the material’s desirability as an anode active material, despite its potential electrochemical advantages.
Natron Energy develops air stable manganese hexacyanomanganate materials treating them with one or more chelating agents. Chelating agents are acid molecules, including oxalic acid , citric acid, and tartaric acid. The chelating agents interact with metal ions on a surface of manganese hexacyanomanganate material. The resultant materials can be represented as NaₘMn[Mn(CN)₆₋ₖLₖ]ⱼ∙(Che)ₕ∙nH₂O, wherein L is a ligand that may be substituted in the place of a CN⁻ ligand and Che is an acid-containing chelating agent. The chelating agent modified manganese hexacyanomanganate materials exhibit diminished reactivity and, consequently, increased stability within the ambient environment, especially oxygen and water, with no appreciable degradation of the desired electrochemical and cycle life performance metrics.
To increase the electrochemical potential of sodium-ion batteries, Natron Energy developed a new type of hexacyanomanganate-based transition metal cyanide coordination compound that is synthesized in methanol. The new hexacyanomanganate-based material has an interstitial methyl alcohol instead of water molecule. The new material used in the fabrication of anode results in an electrochemical reaction potential of -1.3 V vs. the silver/silver-chloride reference electrode (Ag/AgCl, 4 M NaCl). This reaction potential is 0.4 V more negative than the -0.9 V electrochemical reaction potential of previous anode materials synthesized using water. Having a more negative reaction potential is desirable for an anode material because the resulting higher cell voltage results in a greater amount of stored energy.
Innovation of electrolytes
Batteries with prussian blue electrodes may use either aqueous or non-aqueous electrolytes. Aqueous electrolytes would be preferred due to their low cost and superior ionic conductivity. However, using aqueous electrolytes makes it necessary to restrict the design to electrochemical cells with relatively low voltages, typically less than 1.5 V, in order to prevent the electrolytic decomposition of water into hydrogen and oxygen. Therefore, Natron Energy’s batteries with prussian blue electrodes use non-aqueous electrolyte.
The electrolyte typically consists of sodium salts dissolved in an organic solvent such as acetonitrile. Natron Energy developed improved electrolytes by adding a dinitrile material, such as succinonitrile, to increase the battery’s lifespan while allowing for a fast discharge to an extremely high depth of discharge.
These dinitrile molecules inhibit the formation of transition metal oxide precipitates, which degrade cell performance, by interacting with transition metal ions on the surface of prussian blue material. The dinitrile molecules can also suppress the undesirable reactions between nitrile-containing electrolyte solvents and water, which degrades the performance of the batter. Water impurities are difficult to eliminate from prussian blue materials, electrolyte solvents, and electrolyte salts.
How Natron sodium battery works
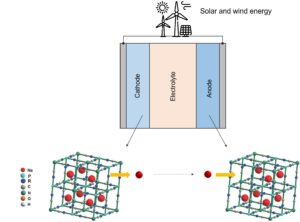
During charging, sodium ions are extracted from the prussian blue cathode and inserted into the prussian blue anode, as depicted in the figure below, while the electrons travel through the external circuit.
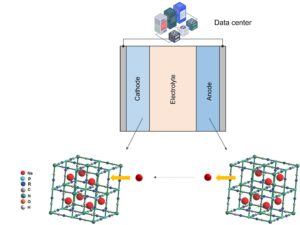
During discharging, the reverse process occurs where the sodium ions are extracted from the anode and reinserted into the cathode, as depicted in the figure below, while the electrons travel through the external circuit performing useful work.
During charging and discharging, the transition metals in the prussian blue undergo redox reactions in conjunction with the insertion or removal of sodium ions. For example, prussian blue material Na₂Mn[Mn(CN)6]∙nH2O contains two redox-active centers of Mn²⁺/Mn³⁺ couples, both of which are capable of undergoing a complete electrochemical redox reaction and contributing a two-electron transfer capacity via a reversible two Na⁺ insertion reaction/extraction process:
Na₂Mn²⁺[Mn²⁺(CN)₆]∙nH₂O ↔ NaMn³⁺[Mn²⁺(CN)₆] + Na⁺ + e⁻
NaMn³⁺[Mn²⁺(CN)₆] ↔ Mn³⁺[Mn³⁺(CN)₆] + Na⁺ + e⁻
The reversible two Na⁺ insertion reaction/extraction in prussian blue materials makes the battery capacity (over 200 mA h g⁻¹) considerably higher than the Na⁺ insertion capacities observed from most of the transition metal oxides (100–150 mA h g⁻¹) and phosphates (≈120 mA h g⁻¹) and is therefore acceptable for practical battery applications.
Natron Energy Patent
- US9876255B2 Cosolvent electrolytes for electrochemical devices
- US9123966B2 Stabilization of battery electrodes using prussian blue analogue coatings
- US10597303B2 Surface-modified cyanide-based transition metal compounds
- US11309594B1 Optimization of electrochemical cell
- US10862168B2 Electrolyte additives for electrochemical devices
- US11056689B2 Transition metal cyanide coordination compounds having enhanced reaction potential
- US11198614B2 Transition metal cyanide coordination compounds having multiple reactions
- US11695163B1 Hierachical structure of transition metal cyanide coordination compounds
- US11728548B1 Separator for electrochemical cell
Natron Sodium Battery Applications
- Data center
Approximately 2% of all global carbon emissions are produced by data centers, which is roughly equivalent to the output of the global airline industry.
The carbon footprint of data centers is affected by electricity consumption, cooling, and location. Data centers require electricity to power their servers, and the location of the data center affects the amount of emissions generated by the data center’s electricity consumption. Data centers can reduce their carbon footprint by implementing energy-efficient cooling practices and utilizing renewable energy sources.
Forced Physics DCT and H5 Data Centers have used Natron’s batteries for their data center operations. Natron’s BlueTray 4000 and Blue Rack™ Sodium-Ion Battery Cabinet provide safe, sustainable backup power for data centers and mission-critical applications.
- Telecom networks
The overall telecommunications (telecom) industry accounts for about 1-3% of global carbon emissions. The telecom industry has recognized the need to reduce its carbon footprint. To do this, the industry has implemented various initiatives, such as optimizing energy consumption and reducing the carbon footprint of the supply chain. Renewable energy sources like solar, wind, and hydropower are being used more and more by telecom companies to power their infrastructure. This shift can reduce their carbon footprint by a lot and make them less dependent on fossil fuels.
Natron Energy offers sodium ion batteries for storing renewable energy to help the telecom industry reduce carbon emissions.
- Industrial mobilities
United Airlines uses Natron’s sodium-ion batteries to power ground service equipment, such as forklifts, tugs, and robots.
- EV fast charging
Chevron uses Natron’s batteries in EV charging stations.
Natron Energy Products
Natron Energy’s batteries are manufactured using commodity materials on existing cell production lines.
Natron BlueTray 4000
As shown in the figure below, the BlueTray 4000 from Natron Energy in a standard 1U 19-inch rackmount configuration delivers 4 kW at 48 V DC over a 2-minute discharge with a 6 kW peak power rating, recharges in 8 minutes, and can cycle >50,000 times.

BlueTray™ 4000 battery is the first sodium-ion battery that achieved a 1973 certification from UL, the definitive third-party evaluation of battery product safety. Natron Energy’s batteries are also the first battery cells to publicly disclose the results of rigorous UL 9540A fire testing on the new UL 9540A database.
Natron Blue Pack
The Natron Blue Pack battery is a 25kW, 48V building block that can be used in systems with voltages up to 812V. For critical power applications at 480 volts, the standard configuration is 10 strings, and for industrial applications at 672 volts, the standard configuration is 14 strings. It is made to charge and discharge very quickly. Depending on the application, it can handle between 50,000 and 100,000 discharge cycles.
This battery is made for uses like Data Centers, UPSs, Telecom, EV Fast Charging, Fuel Cells, Motive/GSE, Industrial Power/Decarbonization, Grid Services – Peak Shaving, Dark Starting, Grid Forming, etc.
Natron Energy Funding
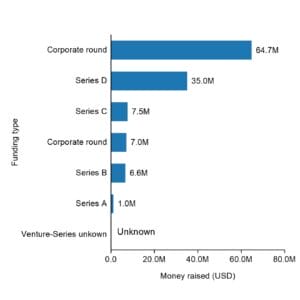
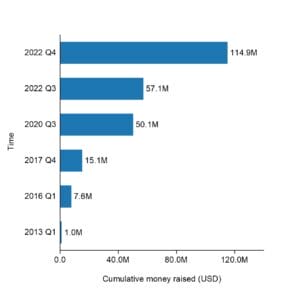
Natron Energy has raised a total of $114.9M in funding over 9 rounds, including
- a Series A round
- a Series B round
- a Series C round
- a Series D round
- two Venture Series-Unknown rounds
- three Corporate rounds
Their latest funding was raised on December 20, 2022 from a Corporate round.
Natron Energy Investors
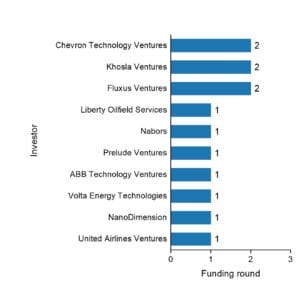
Natron Energy is funded by 10 investors, including
- Khosla Ventures
- Chevron Technology Ventures
- Prelude Ventures
- ABB Technology Ventures
- NanoDimension
- Volta Energy Technologies
- Fluxus Ventures
- Nabors
- Liberty Oilfield Services
- United Airlines Ventures
Liberty Oilfield Services and United Airlines Ventures are the most recent investors.
Natron Energy has a post-money valuation in the range of $100M to $500M as of Jul 21, 2022, according to PrivCo.
Natron Energy Founder
Colin Wessells is Co-Founder.
Natron Energy CEO
Colin Wessells is CEO.
Natron Energy Board Member and Advisor
Joshua Posamentier and Luc Bauer are Board Observers.